Surgical Instrument Tracking with RFID: Preventing Retained Instruments and Saving Thousands of Hours
From autoclavable tags surviving 1,000+ sterilization cycles to Copenhagen's Rigshospitalet projecting 31,000 hours saved annually, RFID surgical instrument tracking is eliminating retained instrument risks and transforming sterile processing departments worldwide.
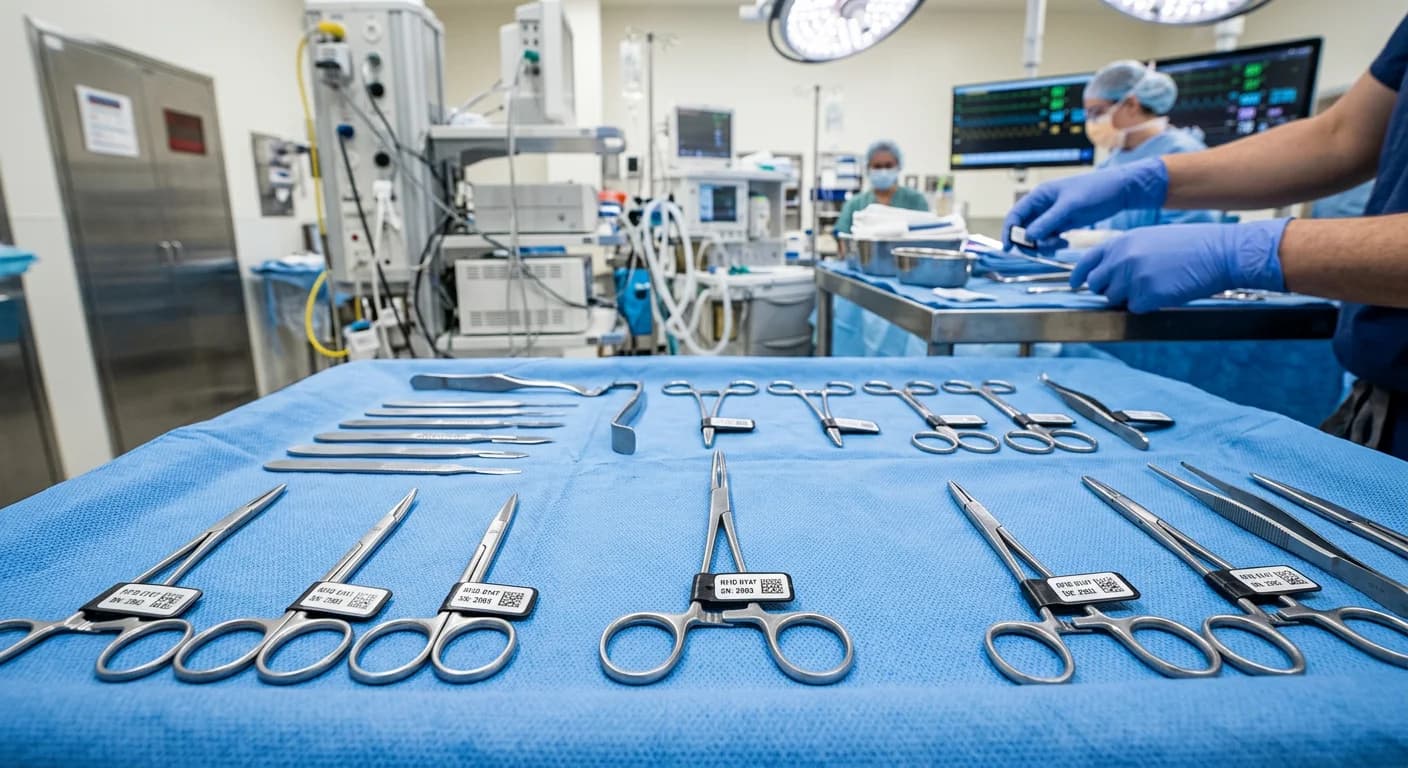
The Retained Surgical Instrument Problem
Retained surgical instruments represent one of the most preventable yet persistent patient safety failures in modern surgery. Despite decades of awareness, manual counting protocols, and regulatory mandates, objects are unintentionally left inside patients at an estimated rate of 1 in every 5,500 surgical procedures in the United States. With approximately 50 million inpatient surgeries performed annually in the US alone, this translates to roughly 9,000 retained surgical instrument cases per year.
The consequences are severe. Retained instruments cause infections, organ perforation, bowel obstruction, and in some cases death. The average cost of a retained instrument incident exceeds $200,000 when combining reoperation costs, extended hospitalization, litigation, and regulatory penalties. Beyond the financial impact, these events represent catastrophic failures of patient trust and institutional credibility.
The root cause is fundamentally a tracking problem. Surgical trays contain 50 to 200 individual instruments, and the standard of care requires manual counting before, during, and after every procedure. Research consistently shows that manual counts have an error rate of 10% to 15%, meaning that one in seven to one in ten counts produces an inaccurate result. When time pressure, staff fatigue, and the complexity of multi-tray procedures compound these counting errors, the conditions for a retained instrument event are created.
RFID technology directly addresses this root cause by providing automated, instantaneous, and error-free instrument identification and counting at every stage of the surgical workflow.
How RFID Surgical Instrument Tracking Works
RFID-based surgical instrument tracking systems employ specially engineered tags, purpose-built readers, and integrated software to create a continuous chain of custody for every instrument from the sterile processing department through the operating room and back.
Autoclavable RFID Tags
The engineering challenge that long delayed RFID adoption in surgical instrument tracking was the sterilization environment. Surgical instruments undergo repeated autoclave cycles at temperatures of 132 to 137 degrees Celsius under high-pressure steam, as well as exposure to harsh chemical sterilants. Standard RFID tags cannot survive these conditions.
Modern surgical instrument RFID tags have overcome this barrier. Constructed from high-temperature ceramics, specialized polymers, and medical-grade adhesives, these tags are designed to withstand more than 1,000 autoclave sterilization cycles without degradation of their RFID functionality. The tags are small enough to be embedded in instrument handles or attached to instrument surfaces without affecting ergonomics or surgical utility.
Leading manufacturers including Xerafy, Tego, and HID Global produce tags specifically engineered for the autoclave environment. These tags use ultra-high frequency RFID operating at 860 to 960 MHz, providing read ranges of up to 2 meters while maintaining reliable identification even when attached to metal surgical instruments, which historically posed challenges for RFID due to signal interference.
Tray-Level vs. Instrument-Level Tracking
RFID surgical instrument tracking can be implemented at two levels of granularity, each offering distinct advantages.
**Tray-level tracking** assigns an RFID tag to each surgical tray as a unit. Readers at sterile processing checkpoints verify that the correct tray is being prepared for the correct procedure, track the tray through the decontamination and sterilization process, and confirm delivery to the appropriate operating room. Tray-level tracking improves workflow efficiency and reduces tray mismatch errors but does not address individual instrument counts.
**Instrument-level tracking** tags every individual instrument within each tray. This provides the full patient safety benefit: automated counting that verifies every instrument is present before the surgical site is closed. When a surgeon requests a final count, a reader scans the entire tray in seconds and immediately identifies any missing instruments. If an instrument is not accounted for, the system alerts the surgical team before the patient leaves the operating room.
Most facilities implementing RFID surgical instrument tracking begin with tray-level tracking for immediate workflow benefits and subsequently transition to instrument-level tracking as the technology matures and tag costs continue to decrease.
Copenhagen Rigshospitalet: A Landmark Case Study
The most comprehensive published evidence for RFID surgical instrument tracking comes from Copenhagen's Rigshospitalet, one of Europe's largest and most advanced university hospitals. In an 18-month pilot study, Rigshospitalet deployed RFID tracking across its surgical instrument processing operations and documented results that have become a reference point for the global healthcare community.
The pilot encompassed tracking of surgical instrument trays through the complete cycle of decontamination, assembly, sterilization, distribution, use, and return. RFID readers were installed at key workflow checkpoints including the decontamination intake, assembly stations, autoclave loading and unloading points, and sterile storage areas.
Projected Savings: 31,000 Hours Per Year
The most striking finding from the Rigshospitalet pilot was the labor efficiency impact. Based on the 18-month study data, the hospital projected that full-scale RFID implementation would save approximately 31,000 staff hours per year in their sterile processing department alone. This figure represents time previously spent on manual tray searches, instrument identification, documentation of processing steps, and resolution of discrepancies.
To put this in perspective, 31,000 hours is equivalent to approximately 15 full-time positions. For a hospital already facing staffing shortages in sterile processing — a challenge common across European and North American healthcare systems — this labor recapture represents both a significant financial saving and a practical solution to workforce constraints.
The pilot also documented a 45% reduction in tray turnaround time, meaning that surgical instrument sets moved from the operating room back to sterile storage significantly faster. This improvement directly reduced the need for duplicate tray sets, which are expensive both to purchase and to maintain.
Error Reduction and Quality Metrics
Beyond efficiency gains, Rigshospitalet reported measurable improvements in quality metrics. Tray assembly errors — cases where incorrect instruments were included in or omitted from surgical trays — decreased by 70%. Processing documentation accuracy improved from 82% to 99%, creating reliable audit trails for quality assurance and regulatory compliance.
The hospital also noted an important secondary benefit: improved staff satisfaction in the sterile processing department. Workers reported that RFID automation reduced the tedious, repetitive aspects of their work and allowed them to focus on tasks requiring clinical judgment and manual skill.
Infection Prevention Through Complete Tracking
RFID surgical instrument tracking contributes to infection prevention in ways that extend beyond the retained instrument problem. Complete instrument tracking ensures that every item passing through the sterile processing department has a verified processing history.
When an instrument's RFID tag is scanned at each processing step — decontamination, ultrasonic cleaning, inspection, assembly, and sterilization — the system creates an unbroken chain of processing evidence. If any step is skipped or performed out of sequence, the system flags the tray as non-compliant before it reaches the operating room.
This capability is particularly valuable during infection outbreak investigations. When a surgical site infection is traced to a potential instrument contamination source, RFID records enable investigators to identify exactly which instruments were used in the affected procedure, which processing cycle they went through, which staff members handled them, and whether any processing anomalies occurred. This level of traceability transforms infection investigation from a weeks-long manual record review into a query that can be answered in minutes.
Hospitals with RFID instrument tracking also report improved sterilizer validation compliance. Because the system automatically records sterilization cycle data for every tagged tray, biological indicator results and chemical integrator readings are linked directly to specific instrument sets rather than relying on manual log entries that may be incomplete or inaccurate.
Regulatory Requirements and the Joint Commission
The Joint Commission, which accredits more than 22,000 healthcare organizations in the United States, has established clear expectations for surgical instrument counting and tracking. The Universal Protocol and National Patient Safety Goals require facilities to perform instrument counts for every surgical procedure and to have policies in place for reconciling count discrepancies.
While the Joint Commission does not mandate any specific technology, their surveyors increasingly recognize RFID systems as evidence of best practice in instrument management. Facilities with automated counting and tracking systems demonstrate a higher level of compliance during accreditation surveys and are better positioned to respond to adverse event investigations with complete documentation.
The European Union Medical Device Regulation further strengthens the case for instrument-level tracking by requiring Unique Device Identification for reusable surgical instruments. RFID tags that encode UDI data satisfy this regulatory requirement while simultaneously providing the operational tracking benefits described above.
Cost-Benefit Analysis
The financial case for RFID surgical instrument tracking is built on multiple cost reduction and cost avoidance factors.
**Direct cost reductions include:** a 30% to 50% decrease in instrument replacement costs as real-time tracking reduces loss and misplacement; a 20% to 40% reduction in duplicate tray sets as faster turnaround times reduce the need for backup inventory; and labor savings equivalent to 10 to 20 full-time employees in sterile processing for a large hospital.
**Cost avoidance includes:** elimination of retained instrument litigation costs averaging $200,000 or more per incident; avoidance of regulatory penalties and accreditation sanctions; and prevention of surgical delays caused by missing or incomplete instrument trays, which cost operating rooms an average of $30 to $60 per minute of downtime.
Implementation costs for a 500-bed hospital typically range from $800,000 to $2 million for the initial deployment, including RFID tags for the existing instrument inventory, readers at processing checkpoints, software licensing, and staff training. Ongoing annual costs for tag replacement and system maintenance range from $150,000 to $400,000.
Based on published data from Rigshospitalet and other implementing facilities, the payback period ranges from 18 to 30 months, with ongoing annual net savings of $500,000 to $1.5 million after the initial investment is recovered. For large academic medical centers with complex surgical programs, the savings can exceed $3 million annually.
Implementation Considerations
Healthcare facilities considering RFID surgical instrument tracking should evaluate several factors before proceeding with deployment.
**Tag selection and attachment:** Not all autoclavable tags are equal. Facilities should require evidence of at least 1,000 sterilization cycle survivability and should conduct pilot testing with their specific autoclave equipment and cycle parameters. Tag attachment method — laser welding, medical-grade adhesive, or mechanical embedding — should be selected based on instrument type and material.
**Reader infrastructure:** RFID readers must be positioned at every critical workflow checkpoint. Portable handheld readers supplement fixed installations by enabling scanning at the surgical field. Reader selection should account for the metal-rich environment of sterile processing departments, where reflections and interference can affect read reliability.
**Software integration:** The RFID system must integrate with the facility's existing instrument tracking software, surgical scheduling system, and quality management platform. Standardized interfaces using GS1 UDI data formats simplify integration and future-proof the investment.
**Change management:** Sterile processing technicians and surgical teams require training not only on the technology itself but on the modified workflows it enables. Successful implementations assign dedicated super-users in each department who serve as first-line support and change champions.
**Phased rollout:** Beginning with high-volume surgical specialties such as orthopedics and general surgery provides the clearest efficiency gains and the largest dataset for validating system performance before expanding to specialized instrument sets in cardiovascular, neurosurgery, and microsurgery.
The Path Forward
The convergence of regulatory pressure, patient safety imperatives, and technology maturation has positioned RFID surgical instrument tracking as an inevitable evolution in operating room and sterile processing standards. The question is no longer whether facilities will adopt this technology but when and how quickly they can deploy it.
As tag costs continue to decline — current per-tag prices of $2 to $5 are projected to fall below $1 within three years — and as the installed base of RFID infrastructure grows, the economic barriers to adoption are rapidly diminishing. Simultaneously, the regulatory and liability risks of relying on manual counting processes are increasing as the availability of proven automated alternatives makes continued reliance on manual methods increasingly difficult to defend.
Healthcare facilities that move early to implement RFID surgical instrument tracking will benefit not only from improved patient safety and operational efficiency but from the institutional knowledge and workflow optimization that comes with early adoption of transformative technology. The evidence from Rigshospitalet and other pioneering institutions is clear: RFID instrument tracking delivers measurable, significant, and sustained improvements in every dimension of surgical instrument management.
Ready to Implement RFID in Your Facility?
Contact us to learn how our RFID solutions can improve patient safety and operational efficiency.
Related Articles
AI-Powered RFID Patient Tracking: The Future of Hospital Safety in 2026
Discover how artificial intelligence combined with RFID technology is revolutionizing patient safety, reducing falls, and preventing wandering incidents in healthcare facilities.

NFC Patient Wristbands: The Future of Bedside Verification
How NFC-enabled wristbands are transforming bedside verification in hospitals, offering tap-to-verify workflows that work through gloves and moisture while integrating seamlessly with smartphones and EHR systems.